Your Microbiome Has a Hormone Problem (And It Goes Both Ways)
Publication 12
Decoding the Interconnected Microbiome
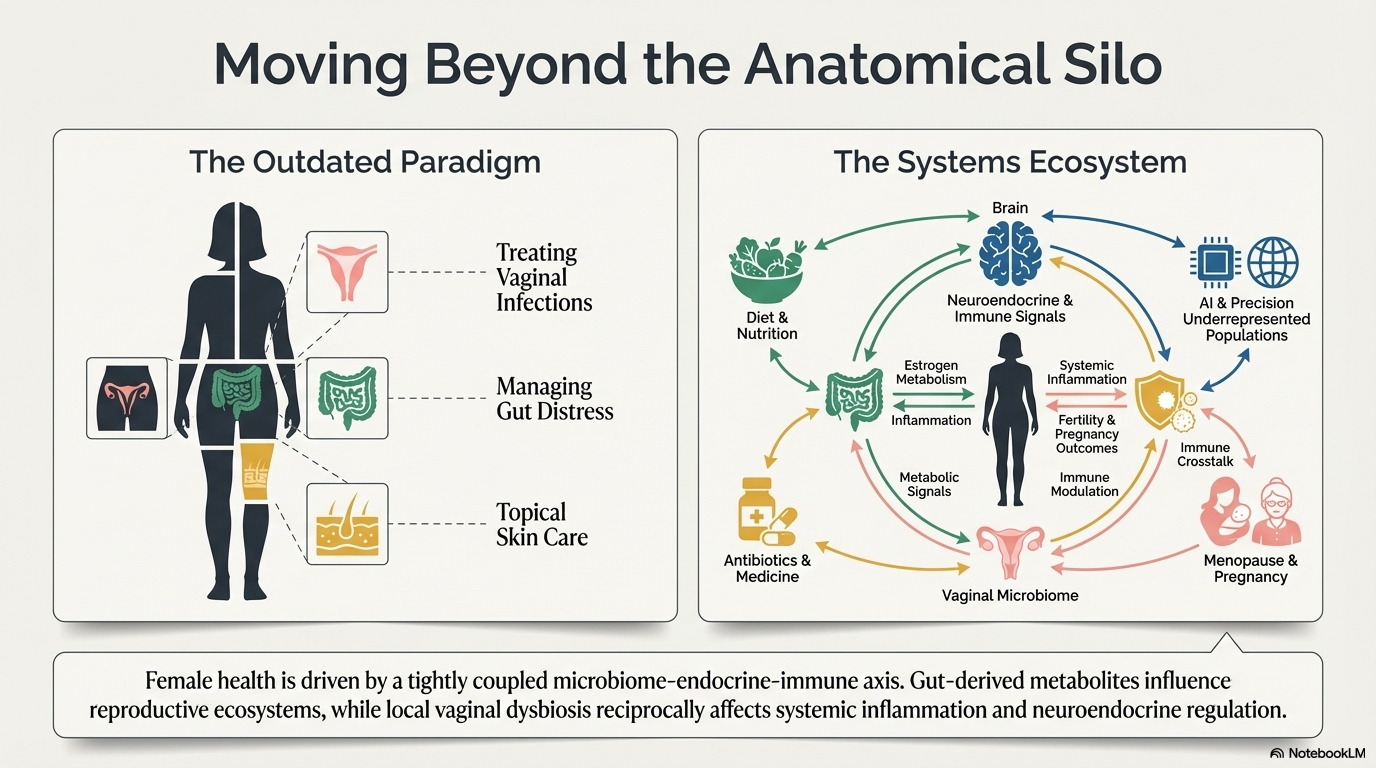
If you've heard of the microbiome, you've probably heard it described as the collection of bacteria living in your gut. That's not wrong, but, it's incomplete — especially if you're a woman.
Women have distinct microbial ecosystems living across multiple body sites: the gut, the vagina, the skin, even breast tissue. And unlike in men, those communities are constantly being shaped — and reshaped — by your hormones. Estrogen, progesterone, cortisol, insulin. They all leave a fingerprint on your microbiome.
But here's the part most people don't know: it's not a one-way street. Your microbiome also shapes your hormones right back.
This is the core of what researchers are now calling the microbiome–endocrine axis, and the evidence for it is growing fast. Two major 2025–2026 reviews — one published in International Journal of Molecular Sciences and one currently in press in Microbiology Research — have put together some of the most comprehensive pictures yet of how this works in women specifically. Let’s dive into the science.
First:
What Is a Microbiome, Really?
Your microbiome is the entire ecosystem of microorganisms living on and inside your body — bacteria, fungi, viruses, and other microbes that co-exist with your cells. The gut gets the most attention, but every body site has its own distinct microbial community with its own composition and its own jobs.
What makes women's microbiomes uniquely complex is hormonal cycling. Fluctuations in estrogen and progesterone across your menstrual cycle, pregnancy, and menopause continuously shift microbial stability, diversity, and which species are present. Men don't experience this kind of ongoing hormonal fluctuation, which is one reason why most of what we know about the microbiome has come from male-dominated research — and why women-specific findings are only now catching up.
The Estrobolome:
Your Gut Controls Your Estrogen Levels
This is the most important concept in women's microbiome research, and it's almost never talked about outside of scientific journals.
Inside your gut lives a subset of bacteria with genes specifically involved in estrogen metabolism. Researchers call this collection the estrobolome. Here's how it works:
Your liver processes estrogen and packages it for removal via bile. That packaged estrogen travels to the gut, where certain bacteria produce an enzyme called beta-glucuronidase. This enzyme cuts the estrogen free from its packaging — essentially recycling it back into active form — so it can re-enter your bloodstream and circulate again.
In other words: your gut bacteria determine how much estrogen is actually active in your body.
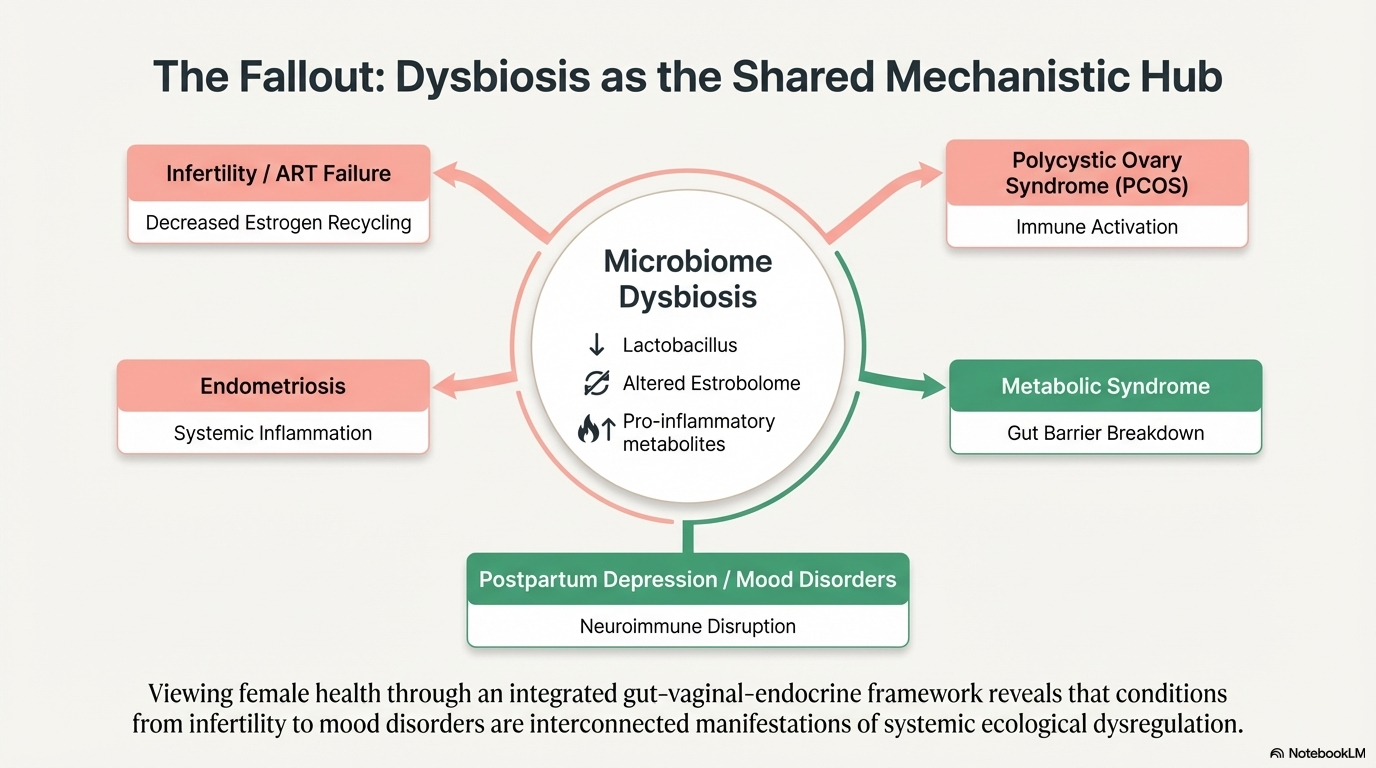
When the estrobolome is balanced, this recycling happens at a healthy rate and your hormone levels stay in check. When the gut microbiome is disrupted — a state called dysbiosis — beta-glucuronidase activity can go haywire. Too much activity means more estrogen gets recycled and estrogen levels climb. Too little means estrogen gets cleared too quickly and levels drop.
Dysregulated estrobolome activity has been linked to conditions including:
Polycystic ovary syndrome (PCOS) — women with PCOS consistently show reduced gut microbial diversity, altered short-chain fatty acid production, and changes in estrobolome-associated enzyme activity
Endometriosis — excessive beta-glucuronidase activity may contribute to estrogen dominance and impaired endometrial receptivity
Breast cancer risk — increased microbial estrogen reactivation has been proposed as a contributor, though this evidence is still largely associative
Menopausal symptoms — even after ovarian estrogen production declines, gut bacteria keep metabolizing estrogen through beta-glucuronidase-producing species
The gut-endocrine connection is also relevant at puberty, when microbial activity may influence reproductive maturation — a window that researchers are only beginning to study seriously.
The Vaginal Microbiome:
Why Lactobacillus Is the One You Want
The vaginal microbiome operates by completely different rules than the gut microbiome. While a diverse gut microbiome is generally considered healthy, the vaginal microbiome is the opposite: less diversity is usually better.
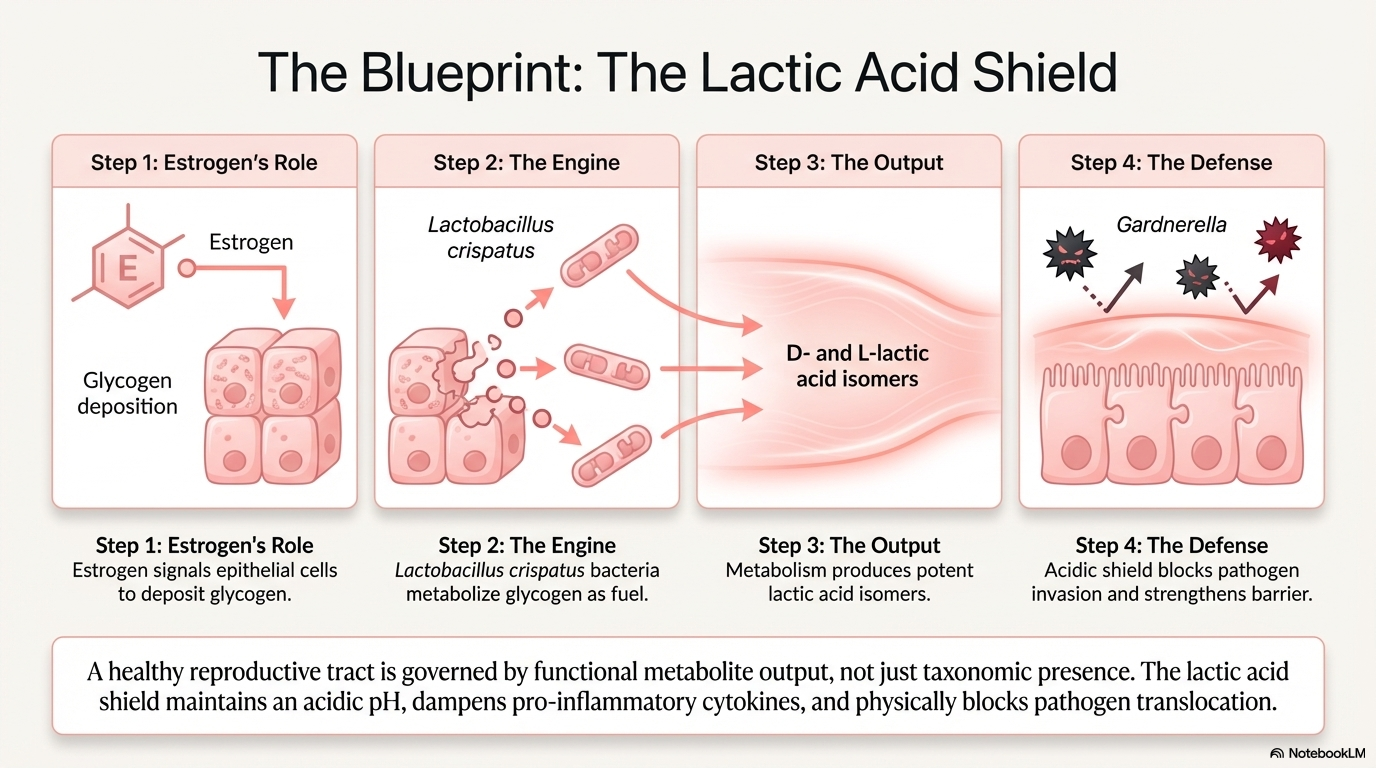
In most reproductive-age women with good vaginal health, the community is dominated by one genus: Lactobacillus. These bacteria produce lactic acid, which keeps vaginal pH low (acidic), and that acidic environment does several important things:
Suppresses the growth of pathogenic bacteria
Reinforces the tight junctions of the vaginal epithelium (essentially strengthening the vaginal wall)
Dampens pro-inflammatory immune responses
Creates a hostile environment for viruses and pathogens
Research using multi-omics approaches (tools that measure microbial metabolites, not just which bacteria are present) has found that the metabolite profile — what bacteria are producing — predicts vaginal health outcomes better than simply knowing which species are there. It's not enough to have Lactobacillus. The species matters, what it's making matters, and the balance matters.
When Lactobacillus declines — due to hormonal shifts, antibiotics, hygiene products, stress, sexual behavior, or other factors — pH rises, the protective barrier weakens, and pro-inflammatory metabolites like succinate accumulate. This is vaginal dysbiosis, and it's strongly associated with:
Bacterial vaginosis (BV) — the most common vaginal infection in reproductive-age women, characterized by overgrowth of species like Gardnerella vaginalis
Increased STI susceptibility — including HIV, HPV persistence, and chlamydia
Fertility problems — more on this in the next section
Adverse pregnancy outcomes
One important nuance: not all Lactobacillus species are equal. Lactobacillus crispatus is considered the gold standard for vaginal health and produces both D- and L-lactic acid. Lactobacillus iners is more common but less protective — it's associated with a less stable microbial environment and may actually transition more easily toward dysbiosis.
The Microbiome and Your Fertility
If you've ever done IVF research, you may have come across the concept of the "endometrial microbiome" and whether it affects implantation. Here's what the current evidence actually shows:
Multiple clinical studies and systematic reviews have found that women with a Lactobacillus-dominant vaginal and cervical microbiome have higher IVF success rates — better implantation rates and higher clinical pregnancy rates — compared to women with dysbiotic profiles. The protective mechanisms are lactic acid-mediated acidification, reduced local inflammation, and better endometrial receptivity.
Conversely, vaginal dysbiosis — particularly overgrowth of anaerobic genera like Gardnerella and Prevotella — has been associated with implantation failure and reduced ART success. Some prospective cohort studies suggest that a non-Lactobacillus-dominant vaginal community can predict pregnancy failure after embryo transfer.
There's also emerging evidence linking gut microbiome dysbiosis to recurrent spontaneous abortion, suggesting the microbiome's influence on reproductive outcomes extends well beyond the vaginal tract.
Worth noting: this research has real methodological limitations. The uterus has a very low microbial biomass and it's hard to distinguish resident microbiota from contamination during sampling. And strain-level differences within Lactobacillus species matter in ways that aren't fully understood yet. This is a promising but still-developing area — not a reason to panic, but worth being aware of.
How Your Hormones Shift the Microbiome Across Your Life
The relationship between hormones and the microbiome isn't static — it changes as your hormonal landscape changes. Here's what the research shows across key life stages:
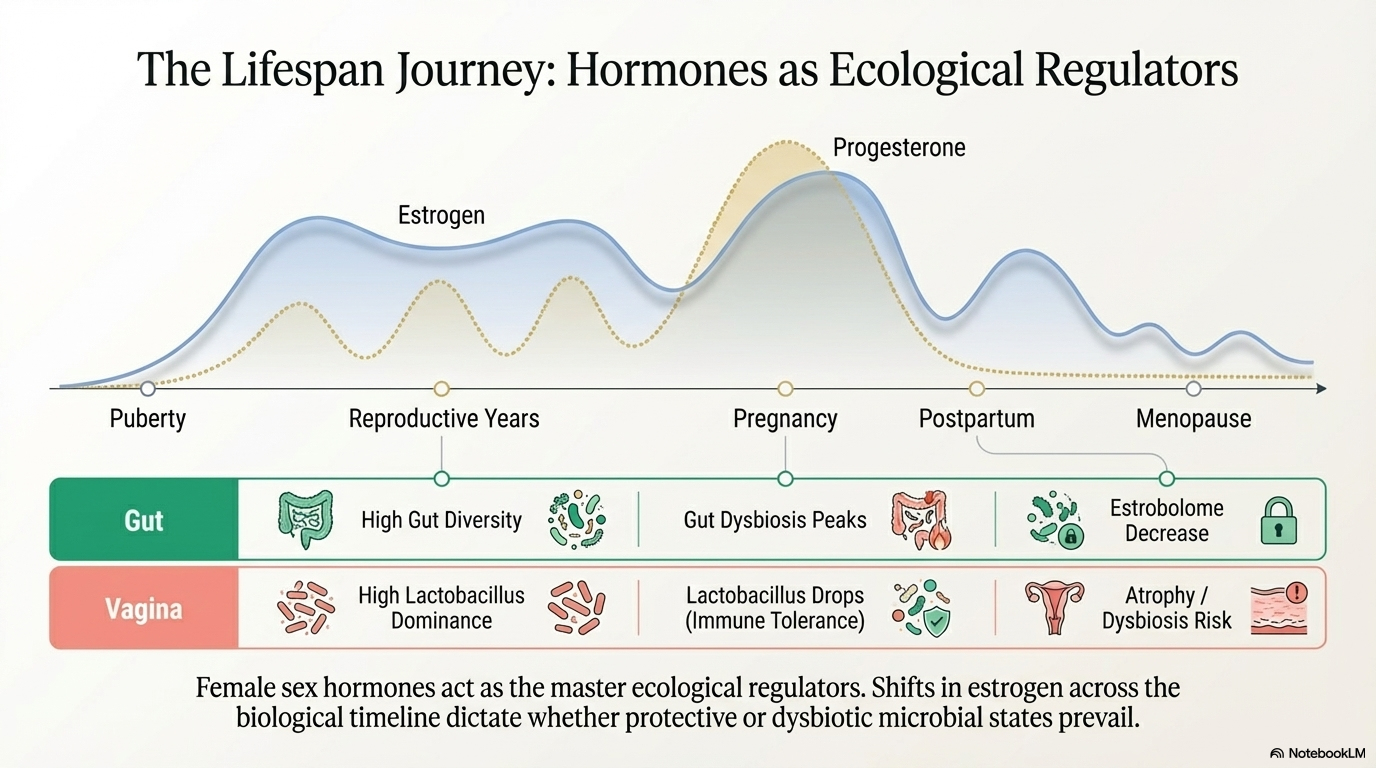
Puberty
As estrogen rises during puberty, the vaginal epithelium starts producing glycogen — a sugar that Lactobacillus feeds on. This is what drives the shift toward a Lactobacillus-dominant vaginal community in adolescence. The gut microbiome also begins to diverge from males at this stage, shaped by rising sex hormones.
The Menstrual Cycle
Estrogen and progesterone fluctuate significantly between the follicular and luteal phases, and research shows corresponding shifts in gut microbial composition across the cycle. This may partly explain why some women notice cyclical digestive changes — bloating, constipation, or diarrhea — that follow their period.
Pregnancy
Pregnancy produces some of the most dramatic microbiome shifts of any life stage. The gut microbiome changes substantially to support maternal immune tolerance, insulin sensitivity, and inflammatory regulation. Alterations in gut microbial composition during pregnancy have been associated with gestational diabetes, pre-eclampsia, and preterm birth.
The vaginal microbiome typically becomes more Lactobacillus-dominant during healthy pregnancy — thought to be a protective adaptation. Disruptions to this pattern are associated with preterm birth risk.
Maternal microbiota may also be transmitted to the infant during and after birth, contributing to fetal immune development and long-term metabolic programming. This is why birth mode (vaginal vs. cesarean) and antibiotic exposure during labor are active areas of research in infant health.
Postpartum
After delivery, estrogen and progesterone drop sharply. The gut microbiome shifts into a dysbiotic state in many women. There's growing evidence of a gut–brain–hormone axis — where microbiome changes interact with neuroimmune and endocrine signaling to influence mood — and this is proposed as one contributing pathway to postpartum depression, though the mechanistic evidence is still being established.
Menopause
As ovarian estrogen production declines, the vaginal microbiome often loses its Lactobacillus dominance. This contributes to vaginal atrophy, dryness, and increased infection susceptibility — common symptoms of menopause that often go undertreated. The gut microbiome shifts too: estrogen metabolism via the estrobolome continues even after menopause, through beta-glucuronidase-producing taxa that influence circulating estrogen levels. This is relevant to both menopausal symptom severity and longer-term disease risk.
The Lifestyle Levers:
What's Actually Shifting Your Microbiome Day to Day
Beyond hormones and life stages, a range of modifiable lifestyle factors meaningfully shape the female microbiome across multiple body sites. This is where the Davidson et al. review is particularly useful — it compiled evidence across the vaginal, gut, oral, and skin microbiomes specifically in women.
Diet
Diet is one of the strongest drivers of gut microbiome composition, and it reaches the vaginal microbiome too — indirectly, through the gut-vagina axis.
What shifts things negatively:
High saturated fat intake is linked to increased BV risk and accumulation of bacterial vaginosis-associated bacteria
Diets high in fat and low in fiber reduce gut microbial diversity and dysregulate estrogen metabolism
Diets deficient in micronutrients (vitamins A, C, D, E, folate, zinc) are associated with vaginal health disruption and increased HPV risk
High glycaemic load diets are linked to increased BV risk
What shifts things positively:
High-fiber diets increase short-chain fatty acid production, which supports gut barrier integrity, immune regulation, and estrogen signaling
Mediterranean and vegetarian dietary patterns are associated with higher microbial diversity
Vitamin D sufficiency is linked to higher Lactobacillus abundance in the vaginal microbiome
Fruit-sourced carbohydrates and plant-based proteins support beneficial gut microbes
Stress
Psychological stress has a direct pathway to vaginal dysbiosis that most people don't know about. Cortisol — your main stress hormone — inhibits glycogen deposition in the vaginal epithelium. Since Lactobacillus feeds on glycogen, less glycogen means less Lactobacillus, which means higher pH and increased dysbiosis risk.
High-stress states have been associated with increased BV incidence, higher likelihood of a dysbiotic vaginal community type, and greater disease severity and persistence for vaginal infections. The gut microbiome is also affected: women under high stress show increased abundance of pro-inflammatory gut bacteria including Escherichia and Shigella.
Smoking
Smoking is a consistent and dose-dependent disruptor of the vaginal microbiome. It suppresses Lactobacillus abundance — likely through anti-estrogenic effects, since estrogen supports the glycogen-Lactobacillus cycle — and is significantly associated with increased BV incidence, higher likelihood of dysbiotic community classification, and increased STI risk including HPV.
Smoking also disrupts the gut microbiome composition and has been identified as an inhibitor of estrogen synthesis through tobacco alkaloids and nicotine — another pathway through which it influences female hormonal health.
Alcohol
Increased alcohol intake is associated with decreased Lactobacillus dominance in the vaginal microbiome and increased BV risk. In the gut, alcohol is linked to dysbiosis, reduced Bacteroidetes, higher Proteobacteria, and systemic inflammation. Effects appear across the vaginal, gut, oral, and skin microbiomes.
Obesity
Obesity has wide-ranging effects on the female microbiome. In the vaginal microbiome, overweight and obese women show reduced Lactobacillus abundance — including lower L. crispatus and higher L. iners — driven in part by reduced estradiol production and impaired glycogen synthesis in the vaginal epithelium. Obese women have a significantly higher incidence of BV.
In the gut, obesity is associated with reduced diversity, a higher Firmicutes/Bacteroidetes ratio, and reduced Bifidobacterium — changes that are thought to be driven more by diet quality than by weight itself.
Physical Activity
Regular exercise is associated with increased vaginal microbial diversity and a decreased risk of transitioning to a dysbiotic vaginal community type. In the gut, exercise is linked to greater alpha-diversity and enrichment of microbes involved in short-chain fatty acid production with immunomodulatory effects.
Hygiene Practices
Vaginal douching is one of the most consistently harmful practices identified in the vaginal microbiome literature. Women who douche are 1.2–1.5 times more likely to develop BV compared to non-douching women, and those who use any feminine hygiene product are 3 times more likely to report adverse vaginal health outcomes. Douching specifically depletes L. crispatus and promotes the growth of dysbiosis-associated species.
Crucially: stopping douching alone is insufficient to restore microbial balance once it's been disrupted. Additional interventions are needed.
Probiotics, Postbiotics, and What the Evidence Actually Supports
Did You Know Probiotic naming works in three levels: genus, species, and strain — for example, Lactobacillus (genus) rhamnosus (species) GG (strain code). The clinical evidence is strain-specific, meaning research on GG tells you nothing about what a different L. rhamnosus strain in a different product will do. If a label doesn't show all three, you don't actually know what you're buying.
The probiotic market makes sweeping claims about gut and vaginal health, and the science is significantly more nuanced than the marketing suggests.
Here's what the research does support:
Vaginal application of L. crispatus has shown the most consistent evidence for promoting vaginal microbiome stability and reducing BV recurrence. Oral probiotics targeting vaginal health have much weaker evidence.
Bifidobacterium animalis and Lactobacillus rhamnosus (at specific strains — always look for the full strain code on the label) have shown benefit for bowel regularity and gastrointestinal symptoms in women. Effects vary significantly by strain, meaning the species name alone is not enough to know whether a product will work.
Probiotic effects cannot be extrapolated across products. If a supplement lists only a genus or species name without a strain code, there is no way to verify whether what's inside has ever been tested for your specific concern.
It's worth noting that even the research reviews this post draws from cite species names without full strain codes — a known gap in probiotic literature that makes cross-study comparisons difficult and is exactly why strain-level transparency on product labels matters.
What the research does not support:
Most commercial probiotics marketed to women are not tested in women-only trials and rely on evidence from male or mixed cohorts
Many products don't disclose strain identity, dosage, or formulation testing
Your body's own healthy vaginal community is not something you can bottle. A probiotic might introduce some Lactobacillus strains and provide some benefit, but it hasn't been proven to restore the full ecosystem
Postbiotics — bioactive metabolites produced by bacteria, like short-chain fatty acids — are an emerging and more promising therapeutic direction. They offer more stability and mechanistic specificity than live bacteria. Early evidence suggests potential benefits in fertility, metabolic regulation, and immune modulation, but women-specific clinical trials are limited.
The honest takeaway: diet-based microbiome support (fiber, polyphenols, fermented foods) has a stronger evidence base for most women than supplement-based interventions.
What to Actually Do With This Information
Based on the current evidence, here are the most supported levers:
For gut and hormonal microbiome health:
Prioritize dietary fiber from diverse plant sources — it feeds the estrobolome-supporting bacteria and SCFA-producing species
Eat polyphenol-rich foods (berries, olive oil, dark leafy greens) — these modulate microbial enzyme activity
Address chronic stress — cortisol disrupts the microbiome through multiple pathways
Limit alcohol — consistently associated with dysbiosis across body sites
For vaginal microbiome health:
Avoid douching and scented vaginal products — the evidence against these is extremely consistent
Eat for your gut — the gut-vagina axis is real, and diet affects the vaginal microbiome indirectly
If you're dealing with recurrent BV, ask your provider about vaginal probiotic formulations containing L. crispatus specifically — this has the strongest evidence
Before buying a probiotic:
Check that the strain is named (genus, species, and ideally strain code — e.g., Lactobacillus rhamnosus GG)
Look for clinical evidence in women for your specific concern
Be skeptical of any supplement making broad "hormonal balance" or "vaginal health" claims without disclosing strain identity and supporting data
Source: Tahtouh Zaatar M, Othman R, Abushawish M, et al. The Women's Microbiome: Molecular Insights, Clinical Gaps, and Future Frontiers in Precision Health with Implications for Gulf Cooperation Council Populations. Int J Mol Sci. 2026;27(6):2521. Published 2026 Mar 10.
Read the full review article here → https://doi.org/10.3390/ijms27062521
Source: Shaping the Female Microbiome: A Review of Lifestyle Factors Influencing the Vaginal, Gut, Oral, and Skin Microenvironments. M Davidson I, Nikbakht E, M O'Neill H, Haupt LM, Dunn PJ. Microb Ecol. Published online March 29, 2026.
Read the full review article here → https://doi.org/10.1007/s00248-026-02747-w