Why Heart Disease Hits Women Differently: What Your X Chromosomes Have to Do With It
Publication 15
The Part of Heart Disease No One Talks to Women About
Heart disease is often thought of as a "man's problem." But women are more likely than men to be misdiagnosed, undertreated, and left out of the research that shapes how doctors understand and treat the disease.
Science has long known that sex hormones play a role — estrogen offering some protection before menopause, testosterone creating certain risks in men. But a new review published in Physiology makes the case that hormones are only part of the story, and that a less-understood piece, the sex chromosomes themselves, and a specific class of genes called "escapee genes" may be contributing to why heart disease looks different in women.
This breakdown explains what that research found, why it matters specifically for women, and what questions it raises for the future of women's heart health.
The Research Question
This was not a new experiment with participants. It was a scientific review — meaning researchers at the Medical College of Georgia synthesized the existing body of evidence on a specific question: beyond hormones, what drives the sex differences we see in cardiovascular disease?
Most of what medicine understands about sex differences in heart disease comes down to hormones — estrogen protecting women before menopause, testosterone creating certain risks in men. But this review argues there's a major missing piece: sex chromosomes themselves, and a specific group of genes that behave unusually in female cells.
The review focused on three interconnected areas:
Sex chromosomes (XX vs. XY): How the genetic difference between male and female cells — beyond just hormones — shapes cardiovascular risk
X-linked "escapee" genes: A subset of genes that behave differently in women's cells compared to men's, and what that means for the heart and blood vessels
Reactive oxygen species (ROS) and oxidative stress: How the balance between cell-damaging molecules and the body's defenses against them differs between sexes, and how this drives conditions like high blood pressure, arterial stiffening, and atherosclerosis
The review also examined specific cardiovascular conditions such as hypertension, arterial stiffening, and atherosclerosis through this lens, and closed with a look at why antioxidant clinical trials have mostly failed.
The Bottom Line:
Women's Bodies Handle Oxidative Stress Differently
At the center of this review is the concept of oxidative stress: what happens when your cells produce more damaging molecules (called reactive oxygen species, or ROS) than your body can neutralize with antioxidants.
Think of it like rust forming on metal. ROS are constantly being produced — that's normal. Your body has antioxidant "rust removers" (like superoxide dismutase, catalase, and glutathione) to keep things in check. When ROS production outpaces those defenses, damage accumulates in blood vessels, the heart, and surrounding tissue.
Before menopause, women are generally better at keeping this balance. Estradiol, the primary form of estrogen, acts as a natural antioxidant, helping blood vessels relax and reducing inflammation. Testosterone, by contrast, tends to promote oxidative stress. This is a significant reason why premenopausal women have lower rates of high blood pressure, arterial stiffening, and atherosclerosis than age-matched men.
But here's where it gets complicated: the hormonal story doesn't explain everything. Even when researchers account for hormones, sex differences in cardiovascular disease remain — which is what led scientists to look more closely at sex chromosomes.
The X Chromosome Factor:
What "Escapee Genes" Are and Why Women Have More of Them
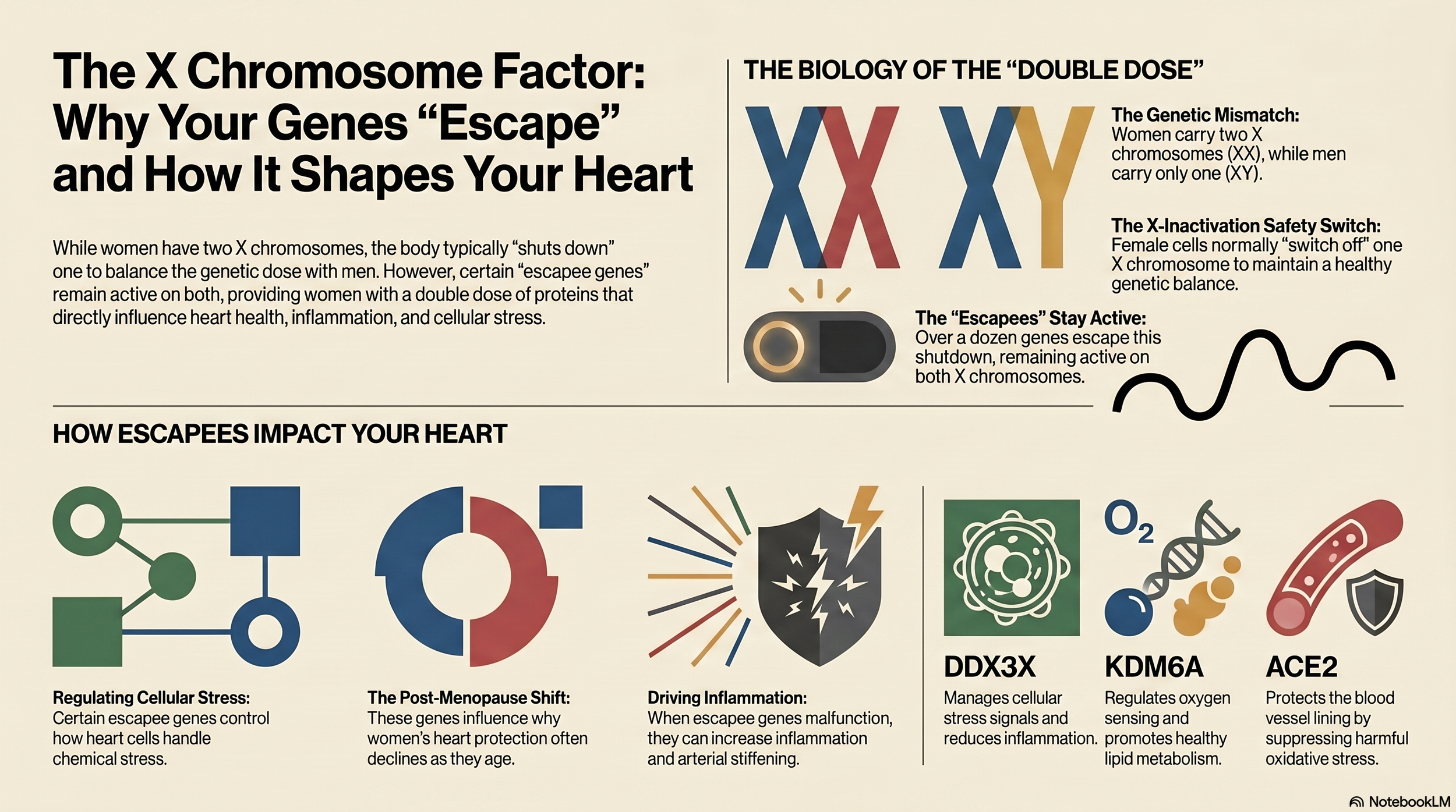
Every woman has two X chromosomes (XX); every man has one X and one Y (XY). To prevent women from having double the dose of X-linked genes, one X chromosome in each female cell is silenced. This process is called X chromosome inactivation (XCI). This happens early in development and is maintained throughout life.
But here's the catch: the silencing isn't complete. About 15% of X-linked genes are never fully turned off. They "escape" inactivation and remain active on both X chromosomes. This means women express higher levels of these specific genes than men do, because men only have one X chromosome to begin with.
These are called escapee genes, and several of them have been shown to directly influence oxidative stress and cardiovascular function:
KDM6A: Regulates how cells sense and respond to low oxygen. In heart tissue, it promotes a damaging type of fat oxidation. During oxygen deprivation, its reduction triggers a form of programmed cell death implicated in CVD. Because women have higher levels of this gene, its role in female cardiovascular disease may be underappreciated.
KDM5C: An epigenetic regulator that, in healthy conditions, helps protect cells from oxidative damage. However, the review notes that in female mice specifically, higher KDM5C activity has been linked to mitochondrial dysfunction in the context of statin treatment — an important nuance for women on statins.
USP9X: A gene involved in cell survival and inflammation. It can activate pathways that promote inflammatory cytokines and increase ROS while reducing antioxidant enzymes. However, the picture is complicated: when USP9X is removed from immune cells called macrophages, atherosclerosis actually worsens — suggesting its role is context-dependent, not simply harmful.
DDX3X: Regulates how cells manage stress. When functioning normally, it helps dampen oxidative stress. When it's deficient, inflammation and immune cell infiltration in tissues increase. Separately, the stress granules that DDX3X regulates have been shown to promote inflammation in atherosclerosis.
BMX & STS: Together, these escapee genes are linked to the activation of myofibroblasts — a cell type involved in tissue stiffening. Research has shown they may contribute specifically to aortic valve stenosis in women, though their connection to oxidative stress pathways is still being established.
ACE2: Unlike the others, this gene is actually more expressed in male cells (particularly in coronary arteries). It plays a protective role by suppressing oxidative enzymes, which helps blood vessels relax. Its relatively lower expression in women may be a piece of why female cardiovascular risk looks different.
How This Plays Out in Three Specific Conditions
High Blood Pressure (Hypertension)
Women's blood pressure is typically lower than men's for much of adulthood, but during the menopausal transition it rises steeply and can ultimately surpass men's. This isn't just about losing estrogen. Research using specialized mouse models where researchers can separate the effects of sex hormones from sex chromosomes shows that having two X chromosomes independently promotes high blood pressure in response to the hormone angiotensin II, regardless of estrogen levels.
This matters because it helps explain why hormone replacement therapy didn't protect all women from cardiovascular events in large clinical trials. The chromosomal contribution exists separately from the hormonal one — and it may require different therapeutic targets.
Arterial Stiffening
As blood vessels lose their elasticity, the heart has to work harder to pump blood through them. This is measured by pulse wave velocity (PWV) — essentially, how fast a pressure wave travels through your arteries. Stiffer arteries = faster wave = higher cardiovascular risk.
Before menopause, women have more elastic arteries than men the same age. But arterial stiffening in women accelerates sharply in the years around menopause. Research has shown that at this stage, oxidative stress is a key driver — and in women specifically, ascorbic acid (Vitamin C) infusions have been shown to reduce arterial stiffness by lowering the activity of oxidative enzymes. This suggests a real, if narrow, window where antioxidant intervention may be helpful for women.
Atherosclerosis (Plaque Buildup)
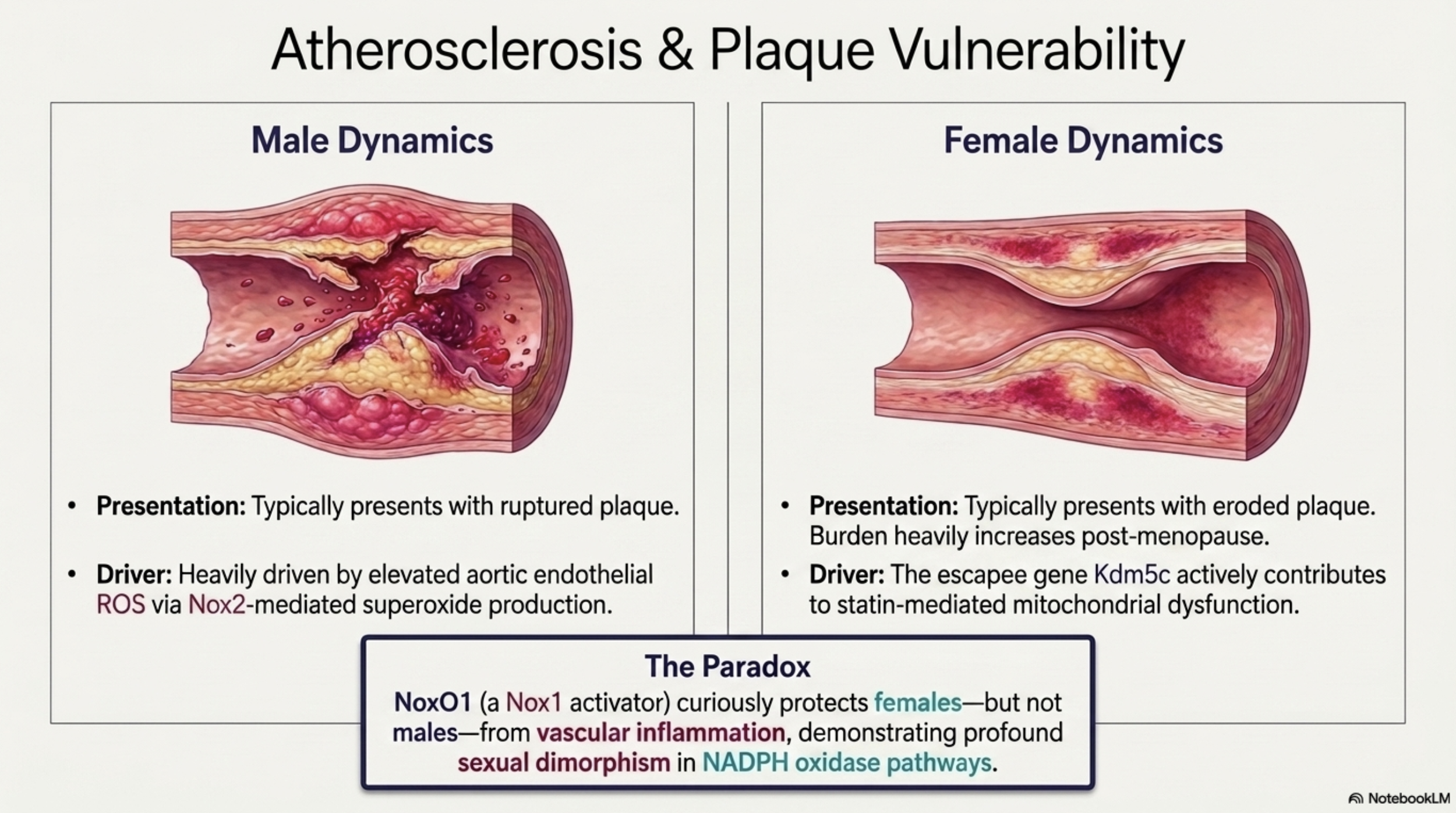
Atherosclerosis, which is the accumulation of plaques in artery walls, presents differently depending on sex:
Men tend to have ruptured plaques (which break open and cause sudden, dramatic blockages)
Women are more likely to have eroded plaques (which wear away more gradually) and women's plaques tend to accumulate more significantly after menopause
This difference in plaque type matters clinically, because the paper notes that women's distinct clinical presentations of CVD influence treatment outcomes — and low awareness of CVD risk in women contributes to delays in diagnosis and treatment. Women are also protected from atherosclerosis before menopause, but after it, their incidence surpasses men's.
At the cellular level, sex differences in oxidative enzyme expression (particularly Nox1, Nox2, and Nox4 in different tissues) help explain why male and female plaques differ in composition and behavior.
Strengths and Limitations:
A Reality Check
What makes this review valuable:
This is a comprehensive synthesis by researchers actively working in vascular biology. It draws together animal studies, human tissue data, genetic research, and clinical trial results into a coherent framework.
Where the evidence is still thin:
The review is transparent about significant knowledge gaps:
Most studies were conducted in male animals or male humans. When sex is included as a variable at all, female-specific findings are often secondary, not the primary focus.
Escapee gene research in women is still early. Most of what we know about genes like KDM6A and KDM5C comes from animal models. Human data in cardiovascular tissue is limited.
The complexity of ROS makes it a hard target. Different oxidative enzymes do different things in different tissues. Nox4, for instance, is protective in blood vessels but harmful in the kidneys. This is part of why antioxidant clinical trials have repeatedly failed — they treat "oxidative stress" as a monolith when it isn't.
No studies on escapee genes in ROS-mediated hypertension in women exist yet. The authors explicitly call this out as a gap.
What This Review Suggests
Your heart disease risk is shaped by more than your hormones. Your sex chromosomes contribute independently to cardiovascular risk.
Menopause is a critical window. The acceleration of blood pressure, arterial stiffening, and atherosclerosis during and after menopause is real, documented, and driven by both hormonal and chromosomal biology. Taking cardiovascular symptoms seriously during perimenopause is important.
Oxidative stress is a key mechanism. The balance between ROS production and antioxidant defense is central to how blood pressure rises, arteries stiffen, and plaques form. The paper specifically notes that regular exercise has been shown to improve mitochondrial efficiency and suppress ROS production, protecting against CVD.
Women may need different treatment targets. If escapee genes contribute to cardiovascular disease in women in ways that are unique to female biology, then treatments developed primarily in male subjects may not work as well — or may work differently — in women. This is an active area of investigation.
What This Review Does Not Show
It is not a clinical trial. No specific treatment was tested. This is a synthesis of existing science, not a recommendation for any drug, supplement, or intervention.
Escapee gene research is not ready for clinical application. Understanding that KDM6A influences oxygen sensing in heart tissue is important science — but there is no established therapy targeting this gene in humans yet.
It doesn't cover all women equally. Most of the underlying studies used animal models or were not designed to capture variation across ethnicity, age range, or health status. Findings may not apply uniformly.
Antioxidant supplements are not a proven solution. Despite the central role of oxidative stress in CVD, clinical trials for Vitamin C, Vitamin E, and various other antioxidants have largely failed to show consistent cardiovascular benefit. The biology is more specific and context-dependent than a general supplement can address.
For women, especially those approaching or navigating menopause, this review is a reminder that heart health deserves the same attention we give to bone density, hormones, and cancer screening.
Source: Okoye DO, Aboagye E, Swenson S, Ngugi S, Shay K, Ogola BO. Sex Differences and Oxidative Stress in Cardiovascular Disease. Physiology (Bethesda). Published online March 24, 2026.
Read the full review article here → https://doi.org/10.1152/physiol.00039.2025