Keto Diet and PCOS: What the Latest 2026 Meta-Analysis Actually Shows
Publication 10
Why Diet is the Frontline for PCOS
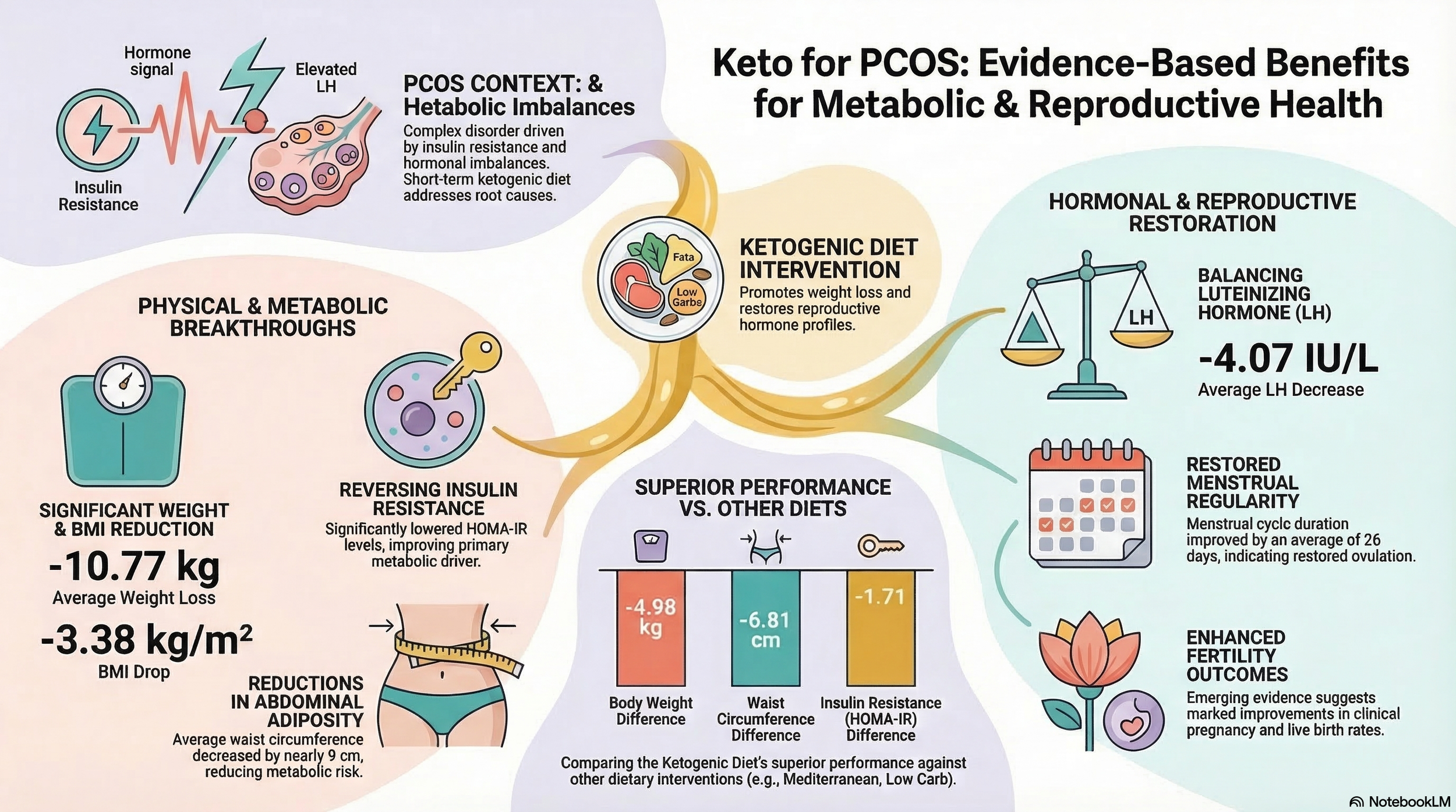
Polycystic Ovary Syndrome (PCOS) is often misunderstood as merely a reproductive issue, but as those living with it know, it is a complex endocrine and metabolic landscape. At its core, PCOS is frequently driven by insulin resistance, which creates a ripple effect: weight gain, hormonal imbalances, and reproductive dysfunction. Because these systems are so deeply intertwined, dietary strategy isn't just about "weight loss" — it's about metabolic repair.
Among the various strategies, the Ketogenic ("Keto") diet has sparked intense debate. To move beyond anecdotes, this review draws on a landmark 2026 meta-analysis published in the journal Clinical Nutrition. This "study of studies" synthesized data to determine whether the metabolic shift into ketosis actually moves the needle on the biological markers that define the PCOS experience.
Research Question & Study Design:
What Was the Mission?
The researchers conducted a systematic review and meta-analysis to evaluate the Ketogenic Diet's (KD) impact. Here is how the study was structured:
Study Type: A systematic review and meta-analysis — a high-level synthesis that pools data from multiple trials to identify broader scientific trends. Importantly, 15 studies qualified for the systematic review, but only 10 met the stricter criteria for inclusion in the quantitative meta-analysis (requiring baseline-vs.-post or active comparator data). Case reports and case series were reviewed narratively but excluded from the statistical pooling. This distinction matters when interpreting the strength of specific findings.
The Goal: To measure the effect of a KD on body weight and composition (anthropometrics), metabolic markers (insulin and lipids), and reproductive hormones.
The Participants: Women with PCOS. Critically, the mean BMI across all included studies exceeded 25 kg/m², anchoring the results specifically to women who are overweight or obese.
The Intervention: A KD defined by low carbohydrate intake (typically under 50g/day, with some studies requiring under 30g/day), high fat, and moderate protein — designed to induce nutritional ketosis.
Duration: Trial durations ranged from approximately 45 days to 8 months, a range that carries meaningful implications for sustainability discussions.
Key Findings:
What the Data Actually Says
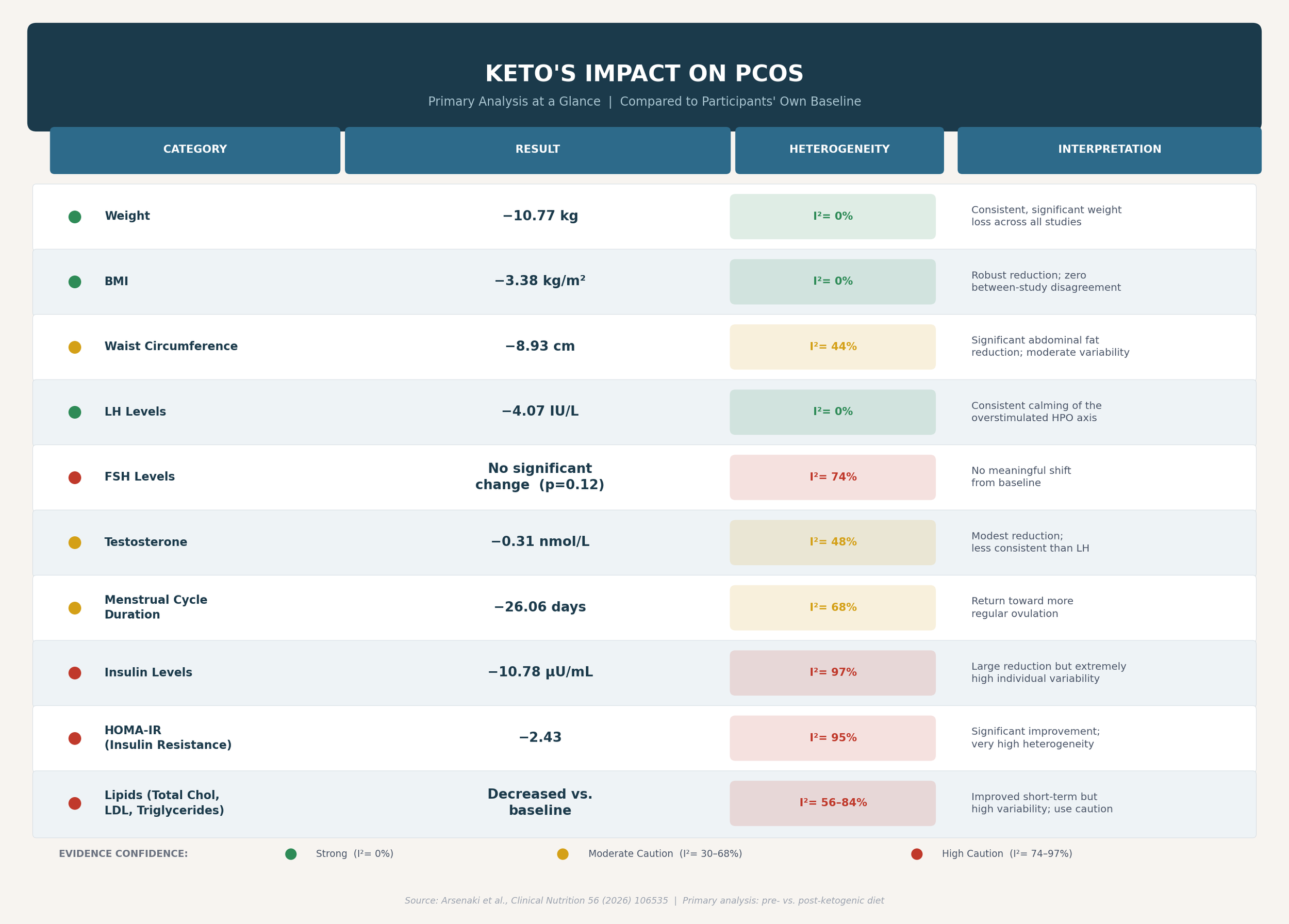
The analysis distinguishes between Primary Findings (how Keto compared to participants' own baseline) and Secondary Findings (how Keto compared to other dietary interventions such as the Mediterranean diet).
Keto vs. Other Healthy Diets:
Secondary Analysis
When compared head-to-head with other dietary interventions, the picture becomes more nuanced:
Keto was superior for: Weight loss (−4.98 kg, I²=7%), BMI (−1.65 kg/m², I²=0%), waist circumference (−6.81 cm, I²=12%), LH reduction (−1.68 IU/L, I²=30%), HOMA-IR (−1.71, I²=90%), and triglycerides (−0.10 mmol/L, I²=9% — this reached statistical significance unlike total cholesterol and LDL).
No significant between-group difference for: Total cholesterol, LDL, HDL, and — critically — testosterone (MD: −0.36 nmol/L, I²=94%; p=0.19). This suggests that while Keto is a standout for weight, abdominal adiposity, and LH management, other balanced dietary approaches may be equally effective for androgen control.
A notable FSH finding: FSH slightly but significantly increased in the keto group relative to other diets (MD: +0.73 IU/L, 95% CI: 0.05–1.40, I²=0%). While not alarming in isolation, this shift in the LH/FSH ratio is a clinically relevant hormonal nuance that warrants attention in future research.
Insulin levels alone (as distinct from HOMA-IR) did not reach statistical significance in the secondary analysis (MD: −6.87 μU/mL, 95% CI: −14.17 to +0.43, I²=88%). The favorable picture for insulin resistance is more robustly supported by HOMA-IR, which is a composite index, than by fasting insulin alone.
Strength of Evidence & The "Heterogeneity" Factor
Evaluating the confidence in these results requires separating two distinct assessments that are sometimes conflated:
I² (Statistical Heterogeneity) measures how much the individual studies disagree with one another in their numerical results. A high I² means the studies produced widely varying effect sizes, reducing confidence that the pooled average reflects a reliable signal.
GRADE (Grading of Recommendations, Assessment, Development and Evaluations) is a broader quality rating that accounts for five factors: risk of bias in individual studies, inconsistency (heterogeneity), indirectness of evidence, imprecision (small sample sizes), and publication bias. An outcome can have low I² but still receive a low GRADE rating if, for example, the total number of participants is small.
Applying both lenses:
Stronger Evidence (Green): Weight loss, BMI reduction, and LH levels. I²=0% across the board — a remarkable degree of cross-study consistency for outcomes of this complexity.
Proceed with Caution (Yellow): Waist circumference (I²=44%), menstrual cycle duration (I²=68%), testosterone (I²=48%), and FSH (I²=74%). Directionally promising but with meaningful between-study variability.
High Caution Required (Red): Insulin levels (I²=97%) and HOMA-IR (I²=95%) in the primary analysis; testosterone (I²=94%) and insulin in the secondary analysis. Results here are highly variable — the "average" improvement may mask individuals who responded poorly or not at all.
The Trial Sequential Analysis caveat: The study applied Trial Sequential Analysis, which revealed that the accumulated sample sizes were generally insufficient to definitively cross statistical benefit or futility thresholds. In plain terms: the data is promising, but the total number of women studied is not yet large enough for scientists to rule out the possibility that some results are partly attributable to chance.
Limitations: The Reality Check
A few caveats deserve direct attention before drawing conclusions:
Who this applies to: Every included study had a mean BMI above 25 kg/m². These findings cannot be extrapolated to Lean PCOS, where different drivers — adrenal hyperandrogenism, HPA axis dysregulation — may not respond the same way, or could be complicated by a high-fat approach.
The time ceiling: Study durations ran from 45 days to 8 months. There is no data from this research on what happens beyond that window. Long-term cardiovascular and metabolic effects remain genuinely unknown.
Study quality: Most included studies were observational cohorts, not RCTs. Of the five RCTs, three had notable risk of bias concerns. Testosterone and gonadotropin measurements were also inconsistently timed, which meaningfully affects the reliability of hormonal results.
The lipid question: Short-term lipid profiles held steady or improved. However, observational data from other populations with follow-up beyond 12 months has flagged increases in LDL and total cholesterol — not a finding of this paper, but a signal worth monitoring.
Tolerability was generally good: Adverse events appeared in only two studies and were mild — dizziness, dehydration, constipation, and transient hypoglycaemia. Dropout was rarely attributed to the diet itself.
Fertility & Reproductive Findings
Two studies outside the quantitative pooling reported reproductive outcomes worth noting:
Rossetti et al. found that after 45 days on keto, median menstrual cycle length dropped from 45 to 32 days (p=0.012), with a significant reduction in ovarian volume (p=0.029) — suggesting restored ovulatory cycling and improved polycystic morphology.
Palafox-Gomez et al. reported striking IVF improvements: implantation rates of 83.3% vs. 8.3%, and clinical pregnancy and live birth rates of 66.7% vs. 0% — with no change in oocyte or embryo quality, pointing to the metabolic environment rather than gamete quality as the mechanism.
These are small, retrospective findings and should not be read as clinical recommendations. The proposed mechanism operates on three levels: abdominal fat loss restores HPO axis signaling; ketosis independently reduces circulating insulin and IGF-1, suppressing androgen overproduction; and rising SHBG binds excess free testosterone as visceral fat is lost. Women in perimenopause and postmenopause were not studied.
What This Study Does Not Prove
Not a cure. It manages metabolic markers — it does not address the genetic or neuroendocrine roots of PCOS.
Not long-term validated. Eight months is the evidence ceiling. Cardiovascular safety beyond that is unknown.
Not universal. The Trial Sequential Analysis found the total sample sizes insufficient to cross definitive benefit thresholds. This is a promising option — not a clinical mandate for every phenotype.
The Takeaway
For women with PCOS and a BMI above 25 kg/m², a short-term ketogenic approach — roughly 6 weeks to 8 months — represents a metabolically meaningful intervention: reducing insulin burden, lowering abdominal adiposity, suppressing excess LH, and potentially restoring ovulatory function. The high heterogeneity in insulin and HOMA-IR outcomes, however, is a direct signal that individual response varies considerably. Keto is one evidence-supported tool — not the definitive one.
What the Research Still Needs
Long-term RCTs beyond 8 months with standardized keto definitions
Lean PCOS trials for women who don't need weight loss
Standardized hormonal assays timed to diurnal rhythms and cycle phase
Diverse cohorts beyond the predominantly European and Asian study populations
Dedicated fertility trials with adequate sample sizes to support clinical guidance
Source: The effects of ketogenic diet on polycystic ovary syndrome: A systematic review and meta-analysis. Arsenaki, E., Stathi, D., Triantafyllidis, K. K., Seldon, Y., Bobotis, S., Lockett, G., Haran, S., Kyrgiou, M., Saso, S., & Kechagias, K. S. (2026). Clinical nutrition (Edinburgh, Scotland), 56, 106535.
Read the full review article here → https://doi.org/10.1016/j.clnu.2025.11.019
Continue Exploring PCOS Research
This article is part of an ongoing research series examining PCOS, hormonal health, metabolism, inflammation, and evidence-based interventions.
Explore the full PCOS research guide:
PCOS Explained: Causes, Symptoms, and Evidence-Based Research Insights